The first time you hear that viruses from space are coming back to Earth, your mind probably jumps straight to science-fiction horror. Silver rockets. Quarantined labs. Panicked scientists in white suits. The truth, as it turns out, is quieter and stranger—and maybe even more beautiful. On a laboratory bench back on Earth, inside clear plastic dishes, something remarkable has been happening. Tiny viruses, sent to the International Space Station, have returned… sharper. More efficient. Better at killing bacteria than when they left. No alien infection, no cosmic monster—just evolution, nudged along by microgravity and the strange physics of orbit.
The Day the Viruses Came Home
Imagine a small, unmarked package arriving at a research lab. No drama, no fanfare. Inside, though, lies the aftermath of months in orbit aboard the space station: vials of clear liquid, each containing billions of bacteriophages—viruses that infect bacteria, not humans. They were sent up as part of an experiment: What happens to viruses in space? Do they change? Do they break down? Do they evolve?
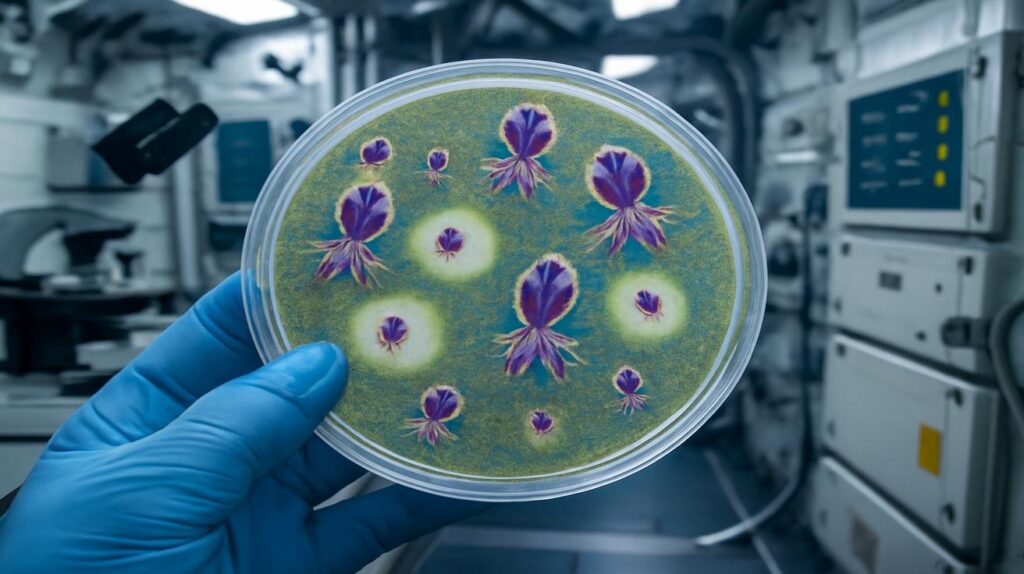
The vials are carefully thawed. Technicians work with the kind of quiet focus that makes a room feel like a chapel—gloved hands, gently clinking glassware, the soft hum of incubators. A scientist draws up a small volume of the space-exposed phages and mixes them with a culture of bacteria on a thin layer of nutrient gel. You wouldn’t notice anything unusual at first. Just a foggy lawn of bacterial growth, like frost spreading over a window. Then, after a little time, the changes appear.
Clear circles begin to blossom across the plate—small zones where bacteria have been killed off by the phages. They’re called plaques, and they look almost like stars punched into a cloudy sky. Except this time, the plaques formed by the “space” viruses are bigger. Cleaner. More aggressive. Side by side with control samples that never left Earth, the difference is subtle but undeniable.
In that quiet lab, the question takes shape: What, exactly, did space do to these viruses?
Microgravity: A Very Strange Garden
Space, as far as life is concerned, is bizarre. On the International Space Station, everything we take for granted on Earth is different—even the way fluids move. There’s almost no gravity to pull liquids down, no settling, no simple convection currents. Instead of sinking or rising, droplets wander. Nutrients don’t spread the way they do on the ground. Tiny organisms, including viruses and bacteria, experience an environment that is less like a gentle lake and more like a snow globe that never quite settles.
For bacteriophages, this strange environment can change the rules of the game. On Earth, a virus has to find a host by diffusion and chance encounters, drifting through liquid until it bumps into a bacterium. In microgravity, how fluid moves—and how cells cluster and float—may alter how often those encounters happen, and which viruses get the upper hand. Only the most efficient phages, the ones that attach faster, invade quicker, and copy themselves better, may thrive in that environment.
It’s like planting seeds in a garden where wind blows from every direction at once, where rain hangs in midair, and where roots never quite know which way is down. Some seeds fail outright. Others, by accident or design, discover new tricks to survive. Over time, the garden doesn’t just look different. It works differently.
That’s the quiet revolution of the space station: it isn’t just a platform for human astronauts; it’s a tiny orbiting test bed where evolution keeps running, just under different rules.
The Unlikely Heroes: Bacteriophages
Bacteriophages—or just “phages”—sound menacing, but their favorite victims are bacteria. Under an electron microscope, they look a little like lunar landers: a geometric head filled with genetic material, perched on spidery legs waiting to grab hold of a cell. Once they latch onto a bacterium, they inject their DNA or RNA inside, hijack the cell’s machinery, and turn it into a viral factory. Eventually, the cell bursts, and new phages spill out.
For decades, phages were scientific curiosities, overshadowed by antibiotics. But now that we’re facing a global crisis of antibiotic resistance, scientists are giving these old viruses a hard new look. Phage therapy—using viruses to treat bacterial infections—is moving from the fringes toward the forefront of experimental medicine.
If you’re trying to design a better, sharper tool against bacteria, you’d want phages that are better at doing what they already do best: infecting and killing their hosts. So when researchers discovered that phages evolved in space were more effective at killing bacteria upon return to Earth, it was like opening a door into a room no one realized existed.
Watching Evolution in Fast-Forward
To understand what changed in these spacefaring viruses, you have to think in generations, not days. A phage can go from infection to explosion of new viral particles in under an hour. Over a single mission, you’re not just sending a static sample to orbit; you’re giving evolution time to play with the dials. Mutation, selection, and survival are still at work, even 400 kilometers above our heads.
In the space station, the phages and their bacterial hosts experience radiation, microgravity, and an altered chemical environment. DNA gets nicked and shuffled by cosmic rays. Proteins fold differently in fluid that doesn’t settle. Bacteria themselves may respond to stress by changing how they express surface molecules—the very features that phages latch onto.
Now imagine that scenario repeating over many generations. Some mutant phages lose the ability to bind to altered bacterial surfaces and quietly vanish from the population. Others acquire a tweak that lets them bind more tightly or inject their genetic material more rapidly. Every small advantage becomes amplified, because the environment keeps pressing. By the time those phages come back to Earth, their lineage is not the same as when they left. The winners are those that learned, through blind trial and error, how to be better killers.
In the lab, this plays out in visible, measurable ways. Plaques from space-evolved phages may spread faster through a bacterial lawn. Researchers can measure higher “burst sizes” (more new viruses produced per infected cell) or shorter latent periods (less time between infection and cell lysis). Under the hood, sequencing their genomes reveals subtle mutations: a swapped base here, a shifted loop in a binding protein there. It doesn’t look like much on a printout, but in the microscopic war zone, these are the differences between a dud round and a precision weapon.
How Scientists Compare Space and Earth Viruses
Back on Earth, comparing space-evolved phages with their ground-based siblings becomes a careful dance of numbers and observations. The experiments might look like this: equal numbers of phages from both groups are added to identical bacterial cultures. The scientists track how quickly the bacteria die, how many phages emerge, and how efficiently each virus can infect at different concentrations.
Those results often end up in tidy spreadsheets and figures, but at their core they tell a simple story: some of the phages that went to space come back “improved” at their job of killing bacteria. To visualize the differences, the data might be laid out in a compact comparison like this:
| Property | Earth‑Evolved Phages | Space‑Evolved Phages |
|---|---|---|
| Time to kill host bacteria | Longer, slower clearance | Shorter, faster clearance |
| Plaque size on culture plates | Smaller, less defined | Larger, more distinct |
| Burst size (viruses per infected cell) | Lower average | Higher average |
| Genetic changes | Baseline sequence | New mutations in key binding regions |
| Potential for phage therapy | Proven but limited by resistance | Promising as enhanced candidates |
These differences don’t mean that all space-altered phages are automatically “better” in every situation. Biology rarely works that cleanly. But they offer a proof of concept: if you change the rules of the environment, you can guide evolution along particular pathways—and end up with tools you didn’t have before.
Why This Matters Down Here on Earth
It’s natural to ask the uneasy question: are we playing with fire by letting viruses evolve in orbit, then bringing them back? In this case, the answer leans heavily toward no—and possibly toward hope.
Bacteriophages are picky eaters. Each type usually targets a narrow range of bacterial species and often specific strains. The phages used in space experiments are selected precisely because they pose no known threat to humans, animals, or plants, and because their host bacteria are safe to handle in the lab. Before they ever leave the ground, the protocols to screen, monitor, and contain them are thorough and strict.
What we gain, however, could be transformative. We are running out of effective antibiotics, and hospitals are already wrestling with stubborn, sometimes lethal infections that shrug off our strongest drugs. Phage therapy offers an enticing alternative: a living, adaptable enemy for our bacterial enemies. But designing or discovering the right phage for a particular pathogen can be slow going.
Now imagine using the International Space Station—or future orbital labs—as evolutionary accelerators. You send up a collection of phages that already have some activity against a dangerous bacteria. You let them dance with their hosts under microgravity, bathing them in the odd physics and radiation of orbit. Later, you bring them down and screen for the lineages that have emerged stronger, faster, or more precisely targeted.
It’s not science fiction. It’s directed evolution, using space itself as part of the toolkit.
From Orbit to Hospital Bed?
Before any space‑evolved phage goes anywhere near a patient, it would face a gauntlet of tests: safety checks, genetic sequencing, animal studies, controlled clinical trials. The bar is high, as it should be. But the idea that some of tomorrow’s medicines could carry a quiet footprint of orbit—that the story of a treatment might include months spent circling Earth—is no longer far‑fetched.
There’s also a poetic symmetry in the idea. For much of human history, the sky has been a place of myth and meaning, a canvas for gods and monsters. Now it’s also becoming a laboratory. Instead of fearing “space germs,” we may find ourselves welcoming space‑sharpened allies against infections that threaten lives in intensive care units and clinics around the world.
The Hidden Ecosystem Above Our Heads
On board the space station, microbes quietly share close quarters with astronauts. They ride up on skin and clothing, in food and water, tucked into the tiny seams of equipment. Some are deliberately introduced for experiments; others arrive as stowaways. Over time, all of them feel the tug—or lack of tug—of microgravity and steady exposure to low‑dose radiation.
We tend to think of the station as a pristine human bubble, but it’s really a floating ecosystem. Bacteria colonize surfaces. Fungi grow in damp corners. Cleanliness is enforced by wipes, filters, and regular sample checks, but life is persistent. In that environment, viruses, including phages, are part of a complex and changing microbial community.
Studying phages in space doesn’t just tell us how to make better antibacterial tools. It also sheds light on a larger question: what does “normal” look like for life off Earth? If we plan to send humans on long missions—to the Moon, to Mars, to stations in deep space—we need to understand how microbes evolve in those conditions. Our bodies, our gut bacteria, and the invisible cloud of life that surrounds us will all be part of any extended trip into the cosmos.
Space‑evolved phages are, in a sense, early ambassadors from that microbial future. They carry data, not in the form of neatly typed reports, but encoded in the subtle bends and twists of their DNA.
The Emotional Weight of Tiny Things
It’s easy to lose the thread of wonder in a story like this and let it dissolve into technicalities about burst sizes and plaque assays. But pause for a moment on the image: a glass vial returning from space, carrying an invisible lineage of viruses that have—without intention, without thought—learned to be different because their world changed.
The astronauts who packed that vial into a re‑entry capsule probably didn’t feel the weight of what it contained. To them it was one more labeled container in a long list of items being carefully stowed. Yet in that vial lived the outcome of countless viral generations, each one subject to the quiet editing of microgravity and cosmic radiation.
There’s something humbling in that. The same physical laws that guide the orbits of planets also nudge the evolution of things so small we can’t see them. Send a virus to space, and you change how it moves, how it encounters its host, and how its descendants will behave when they return. The boundary between “Earth life” and “space life” becomes a little blurrier.
The Stories We Tell About Risk and Wonder
Whenever something biological crosses the line between Earth and space, our imaginations tend to swing between awe and alarm. We picture either miraculous cures or unintended plagues. The reality is often quieter, but no less profound. The experiment with space‑evolved phages sits right in that middle ground: a careful, contained test that reveals both possibility and responsibility.
On the one hand, we’re learning that evolution can be steered using environments beyond our planet, opening potential paths to new therapies. On the other hand, we’re reminded that life adapts quickly—and that every new frontier demands deeper understanding before we scale it up.
In the years to come, we may see more missions sending microbial communities, phage libraries, and even early prototypes of medical tools into orbit. Future astronauts might not only conduct experiments but also rely on treatments that trace their origins to space-based evolution. Perhaps one day, a doctor will stand at a bedside, hanging a bag of fluid that contains phages whose ancestors spun silently around Earth, becoming just a little better at killing a deadly bacterium.
The patient may never hear that story. But it will be there, in the background: a ghost of microgravity, encoded in the invisible architecture of a tiny, efficient virus.
Frequently Asked Questions
Are these space‑evolved viruses dangerous to humans?
The phages used in these experiments are chosen specifically because they infect only bacteria, not human cells. Extensive screening and containment protocols are in place before, during, and after their time in space. Current evidence indicates that they pose no direct threat to people.
How can microgravity make viruses better at killing bacteria?
Microgravity alters how fluids move and how cells and molecules interact. Over many generations, these changes can favor phages that bind more effectively, replicate faster, or produce more offspring. Natural selection in that unusual environment can sharpen certain traits that make them more efficient killers of bacteria.
Could space‑evolved phages replace antibiotics?
They are unlikely to replace antibiotics entirely, but they could become powerful partners. Phage therapy may be especially useful against antibiotic‑resistant infections. Space‑guided evolution could help scientists discover or refine phages that work better against specific problematic bacteria.
Is there a risk of accidentally creating a harmful supervirus in space?
The experiments are designed to minimize that risk. Researchers work with highly specific phages and non‑pathogenic bacterial strains, and they closely monitor genetic and functional changes. Regulatory oversight and biosafety guidelines further reduce the chance of creating anything that could harm humans or the environment.
Why not just evolve phages in special labs on Earth?
Earth‑based directed evolution is already widely used, and it’s extremely valuable. However, space provides a unique environment—microgravity, altered fluid dynamics, and constant low‑dose radiation—that can push evolution along pathways not easily replicated on the ground. It doesn’t replace Earth labs; it adds a new dimension to what’s possible.